Air pollution is created by enhanced concentrations of particles in the air. Some of these particles are so large that you can easily see them, such as dust or sand. However many are much smaller so that they can’t be seen with the naked eye. This fine particulate matter (PM2.5) is often more dangerous because smaller particles can penetrate deeper into the lung. In addition, these particles play an important role in our climate system. In the atmosphere, for example, they absorb and reflect light, and act as condensation nuclei for clouds. Thus PM2.5 plays a key role for public health and for climate change.
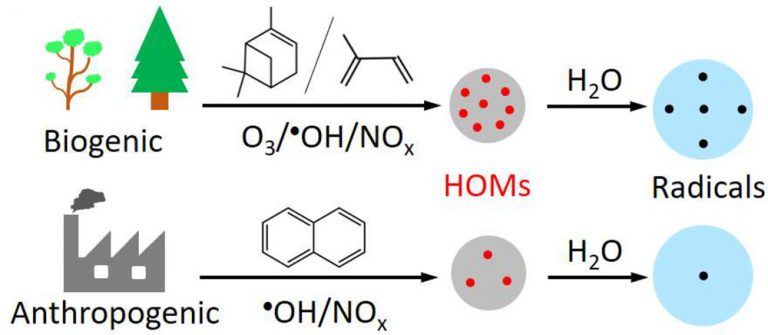
In a new study, Dr. Haijie Tong and co-authors studied a subset of PM2.5, the so-called highly oxygenated molecules (HOMs) and its relationship with radical yield and aerosol oxidative potential. They analyzed fine particulate matter in the air in multiple locations. This including the highly polluted megacity Bejing and in the pristine rainforest at ATTO. They wanted to get insights into the chemical characteristic and evolutions of these HOM particles. In particular, they wanted to find out more about the potential of HOMs to form free radicals. These are highly reactive species with unpaired electrons.
Similar articles
A hidden mechanism continuously forms new aerosol particles from gaseous precursors in the Amazon rainforest, challenging previous assumptions about the sources of cloud condensation nuclei and their role in the hydrological cycle and climate.
New study by the Max Planck Institute for Chemistry shows: The ratio of certain forest scent molecules provides precise insights into the stress state of the rainforest.
Atmospheric aerosol particles are essential for the formation of clouds and precipitation, thereby influencing the Earth’s energy budget, water cycle, and climate. However, the origin of aerosol particles in pristine air over the Amazon rainforest during the wet season is poorly understood. A new study reveals that rainfall regularly induces bursts of newly formed nanoparticles in the air above the forest canopy.
More soot particles enter the central Amazon rainforest from brush fires in Africa than from regional fires at certain times.
In a new study, Marco A. Franco and his colleagues analyzed when and under what conditions aerosols grow to a size relevant for cloud formation. Such growth events are relatively rare in the Amazon rainforest and follow and pronounced diurnal and seasonal cycles. The majority take place during the daytime, and during the wet season. But the team also discovered a few remarkable exceptions.
It is long known that aerosols, directly and indirectly, affect clouds and precipitation. But very few studies have focused on the opposite: the question of how clouds modify aerosol properties. Therefore, Luiz Machado and his colleagues looked into this process at ATTO. Specifically, they studied how weather events influenced the size distribution of aerosol particles.
The Amazon rain forest plays a major role in global hydrological cycling. Biogenic aerosols, such as pollen, fungi, and spores likely influence the formation of clouds and precipitation. However, there are many different types of bioaerosols. The particles vary considerably in size, morphology, mixing state, as well as behavior like hygroscopicity (how much particles attract water) and metabolic activity. Therefore, it is likely that not only the amount of bioaerosols affects the hydrological cycle, but also the types of aerosols present.
Ramsay et al. measured inorganic trace gases such as ammonia and nitric acid and aerosols in the dry season at ATTO. They are to serve as baseline values for their concentration and fluxes in the atmosphere and are a first step in deciphering exchange processes of inorganic trace gases between the Amazon rainforest and the atmosphere.
Soot and other aerosols from biomass burning can influence regional and global weather and climate. Lixia Liu and her colleagues studied how this affects the Amazon Basin during the dry season. While there are many different interactions between biomass burning aerosols and climate, they found that they overall lead to fewer and weaker rain events in the Amazon rainforest.
We hear a lot about particulate matter these days, mostly in the context of air pollution in inner cities. But what about particulate matter in the Amazon rainforest? Well, the short answer is that particulate matter is present in the air above the Amazon, too. And although its concentrations are lower than those in large cities, urbanization and deforestation fires have a significant impact. To find out what that impact exactly is, was the aim of a new study by Suzane de Sá and co-authors.